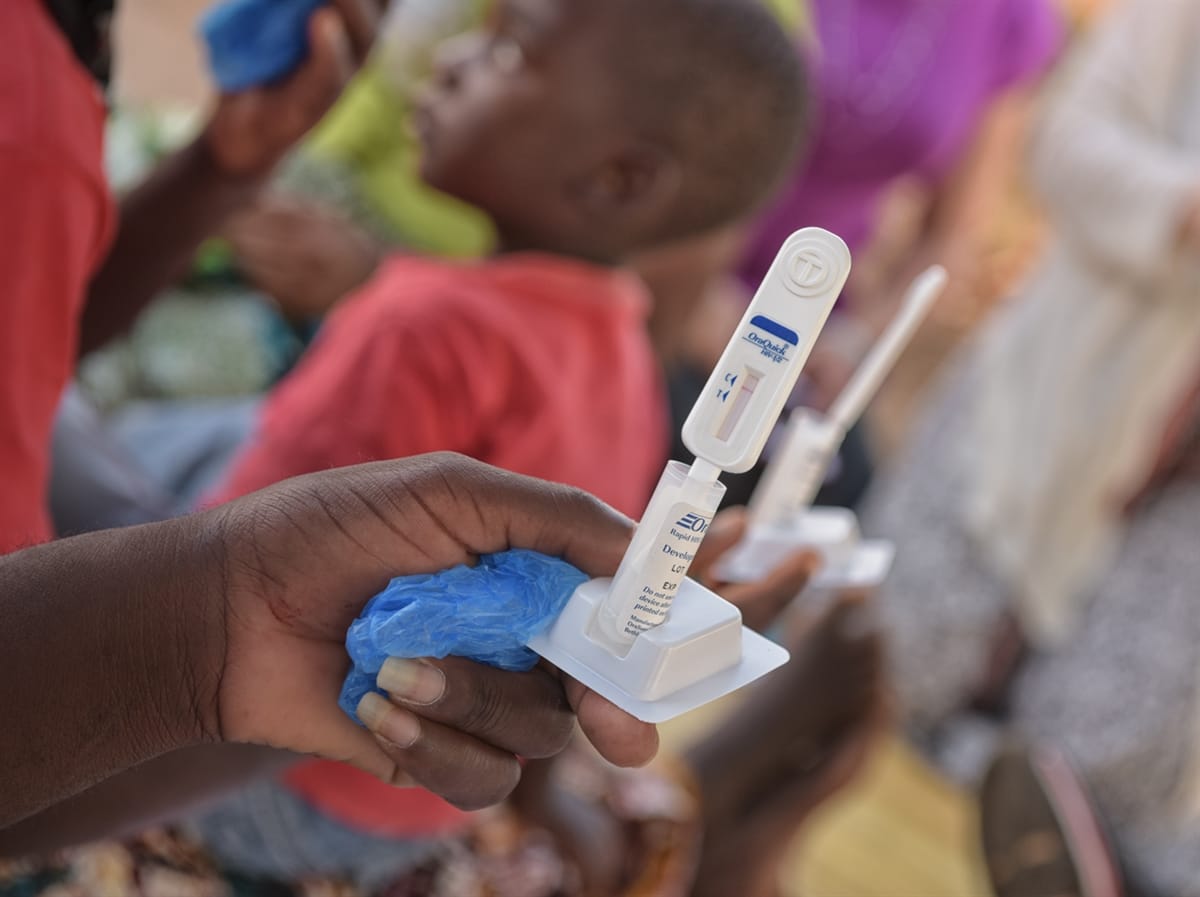
Imagine the first AIDs Free Generation.
At PSI, our team reimagines health care at this scale every day. That’s why we’re launching the first and only rapid HIV self-testing product approved by the World Health Organization. We’re already piloting the project in Malawi, Zambia, South Africa, Lesotho, Swaziland and Zimbabwe.
Making sure all people living with HIV know their status is crucial to slow and eventually stop the virus. But barriers such as stigma and traveling long distances to clinics prevent people from getting tested and then receiving treatment if they test positive.
The self-test — developed by OraSure — is the first and only rapid HIV self-testing product to earn prequalification status by WHO, guaranteeing its quality and safety. This prequalification status enables PSI to access funding for the product so it can be marketed at an affordable price in developing countries.
To date, our program has distributed 485,000 self-test kits, and plans to distribute a total of 4.8 million by the end of the five-year project.
Three out of every 10 people living with HIV do not know they are infected with the virus. The HIV Self-Testing Africa (STAR) Initiative, funded by Unitaid, has amassed compelling evidence that self-testing can reach more people than traditional diagnostics, enabling individuals to learn their HIV status when and where they chose, and seek the treatment they need.
This bold new approach to diagnostics can help meet the United Nation’s ambitious 90-90-90 treatment targets, which call for 90 per cent of people living with HIV to be diagnosed, 90 per cent of those diagnosed to be on treatment and 90 per cent of those on treatment to be virally suppressed by 2020.
Today at the 9th IAS Conference on HIV Science (IAS 2017), the STAR consortium announced the expansion of the HIV Self-Testing Africa project to South Africa, Lesotho and Swaziland, making it the largest effort to date to create a thriving market for HIV self-testing in Africa.
Poor testing coverage among men, young people and other vulnerable populations in sub-Saharan Africa remains a challenge facing the HIV response. HIV self-testing (HIVST), which allows individuals to test themselves in complete privacy, has proven a critical tool to increase the adoption of HIV prevention and treatment services.
The first phase of the STAR project (2015-2017) established that HIVST can be used accurately by almost anyone; is widely accepted when offered through community- and health facility-based distribution models; and can satisfy demand among vulnerable and key populations that do not otherwise use conventional testing services.

“Preliminary results from STAR indicate that self-tests are helping to close knowledge-of-status gaps for groups that have traditionally been hard to reach with other HIV testing services, particularly young people and men,” said STAR Initiative Director Karin Hatzold. “More than a quarter of these populations using self-test kits have never tested before.”
OraQuick HIV self-test kits, manufactured by OraSure Technologies, were used for the first phase of the STAR project. The test allows an individual, using an oral swab, to get a result in as little as 20 minutes. OraQuick is the first HIV rapid diagnostic test prequalified by WHO, a status that indicates it complies with international standards. In addition, the Bill and Melinda Gates Foundation recently announced an agreement to support the affordable sale of OraQuick tests in 50 countries in Africa and Asia.
HIVST volumes generated by the Initiative will improve the health of the global market. HIVST demand will be increasingly predictable, lowering prices and encouraging introduction and scale-up of all available oral- and blood-based tests. This will advance efforts to build a sustainable supply of multiple, quality-assured and affordable HIV self-test products.
Across the three initial STAR countries – Malawi, Zambia and Zimbabwe – 60 to 90 per cent of individuals offered HIVST in rural communities took up the test. Those testing HIV-positive were referred successfully to care and treatment services. In Zimbabwe, 80 per cent of HIV-positive male self-testers reported that they had been referred to care services after self-testing. HIVST also increased the likelihood for HIV-negative men to take up prevention services such as voluntary medical male circumcision (VMMC).
“Thanks to the emerging evidence from STAR and research conducted in other countries, WHO released updated guidelines on HIV self-testing and partner notification in December 2016,” said WHO Technical Officer Cheryl Johnson. “The guidelines strongly recommend that HIVST should be offered as an additional approach to conventional HIV testing services.”

The second phase of the five-year STAR Initiative will build on this success, and aim to demonstrate increased efficiency and potential cost savings of HIVST. About 4.8 million HIV self-test kits will be distributed by 2020. Scaling up HIVST to meet growing demand will increase testing coverage among men, young people and vulnerable groups not currently accessing testing services. As a result, the initiative will directly feed into reaching the UN’s 2020 treatment target. The successful scale-up of HIVST in initial STAR countries and its rapid introduction in new countries under the Initiative will provide a model for adoption in other regions.
“If we are to achieve the first AIDS-free generation, more countries need access to this simple technology, including effective strategies for bringing it to scale,” said PSI President and CEO Karl Hofmann. “Innovative solutions like HIV self-testing helps bring care closer to the people who need it most. When we give consumers simple and cost-effective tools that allow them to take control of their own health we see better outcomes.”
“There is an urgent need to accelerate access to HIV self-testing and increase diagnosis among first-time testers who would not otherwise access testing services,” said Unitaid Executive Director Lelio Mamora. “By overcoming HIVST market constraints and increasing the number of people who know their HIV status, STAR has the potential to turn the course of the epidemic.”
The new five-year STAR project is being implemented by Population Services International (PSI) and Society for Family Health (SFH) South Africa, in collaboration with the World Health Organization (WHO), and consortium partners London School of Hygiene & Tropical Medicine and Wits Reproductive Health & HIV Institute.
About the STAR Consortium
PSI and SFH South Africa head the STAR Initiative, in collaboration with the World Health Organization and MOHs in the six STAR countries. Research activities are led by the London School of Hygiene and Tropical Medicine and their partners Liverpool School of Tropical Medicine, and University College London. PSI leads the consortium Malawi, Zambia, Zimbabwe, Lesotho and Swaziland; in-country research activities are implemented by local research institutions: Malawi-Liverpool-Wellcome Trust Clinical Research Programme, ZAMBART, the Centre for Sexual Health and HIV/AIDS Research Zimbabwe and Africa Health Research Institute. SFH will lead implementation in South Africa with consortium partner University of Witwatersrand-Reproductive Health and HIV Institute. Learn more at www.psiimpact.com/star-hiv-self-testing-africa and www.hivstar.lshtm.ac.uk.